James MFM, Michell WL, Joubert IA, Nicol AJ, Navsaria PH, Gillespie RS. Resuscitation with hydroxyethyl starch improves renal function and lactate clearance in penetrating trauma in a randomized controlled study: the FIRST trial (Fluids in Resuscitation of Severe Trauma). Br J Anaesth. 2011 Nov.;107(5):693–702. PMID 21857015
I remember when I worked in ICU, reading a packet of some colloid and finding out that it was made from Swiss cow’s hooves. Slightly off putting. Better than the tPA made from chinese hamster ovary cells. Mmmm… Chinese hamster ovary cells…
Anyhow.
Cliff Reid reviewed this paper briefly a while back so be sure and read his (more cogent) thoughts on this first.
However, this paper is a classic of what has previously been called wandering outcomes. Though in this one there’s no problems with the registration, more in the writing and conclusions drawn.
METHODS
- Blinded RCT pitting Saline against Hydroxyl Ethyl Starch
- Sponsored by the makers and all the authors have links to industry
- trauma patients, both penetrating and blunt and were analysed separately
- inclusion criteria included a 3L fluid resus, though it’s not clear what they meant by this. They did say that they weren’t allowed more than 2L crystalloid before randomisation
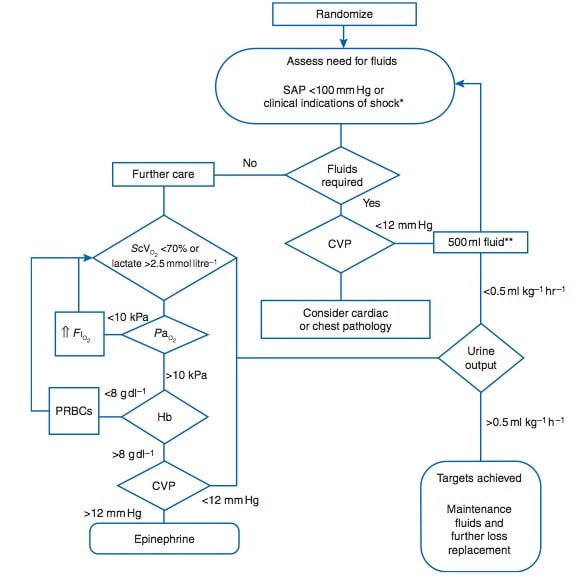
- Below is a little snapshot of their resus algorithm
I just want to point out that that’s a fairly complex protocol which could be questioned at many different points:
- Dutton et al and I’m pretty sure EMCrit would agree that the appropriate fluid to replace in trauma is blood
- the CVP is a poor guide for this
- and in trauma resus do you really wait for the Hb to come back before transfusion?
You get the point that this isn’t crazy-sick-seat-of-your-pants resus but likely a little bit more of a slow controlled environment and probably not the way that I would practice. Though I’d be interested to hear your thoughts on whether or not this is a dodgy algorithm.
RESULTS
- 109 patients split over 4 groups (penetrating v blunt; v HES v saline)
- the SAFE study (the best one on colloids v crystalloids) had 7000 patients
- HES patients were quite a bit sicker
- HES patients needed double the amount of blood
- mortality 16.5% and they say there were no differences statistically but I can’t find the actually mortality figures in the paper. In a study this size 10% v 20% would probably not be statistically significant…
- But none of that is the best bit. Here are the primary outcomes:
Primary outcome variables were the volumes of FIRST fluid needed in the first 24 h after enrolment and the number of patients achieving normal gastrointestinal function by day 5.
Now there’s 2 of them which is a bit naughty but we can live with that. But here are the conclusions in the paper (not even the abstract)
In conclusion, this randomized, controlled, double-blind study has demonstrated faster lactate clearance in penetrating trauma with the use of HES 130/0.4 compared with 0.9% saline without clinically relevant coagulopathy. The superior resuscitation had an outcome benefit, in that no HES patients demonstrated renal injury compared with an incidence of 16% in the saline group. No advantage could be shown for HES in blunt trauma
Now technically all of their conclusion is true (though you could argue about the “outcome benefit” that they claim) but the key point is that in neither the abstract, nor manuscript conclusions do the authors mention the primary outcome. In other words the title, abstract and conclusions give the impression that this was a +ve trial when in fact it was strikingly -ve.
The primary outcome (on which the whole trial is predicated) becomes a side issue to the surrogate outcomes.
Sigh…

Pingback: R&R In The FASTLANE 010 • LITFL • Research and Reviews