As adapted from a recent local teaching session I prepared.
As a broad topic it appears as a common textbook chapter in EM textbooks and there’s a number of nice #FOAMed posts on the topic (referenced below)
Neutropenic Sepsis
The most common, and I would say the most important one. We a lot of this in pretty much every department I’ve worked in. Because it’s so well recognised it should have almost a knee jerk, protocolised response.
In a nut shell, many chemotherapy agents suppress the bone marrow and suppress neutrophil production (amongst other things). Radiotherapy can do it as well but quite uncommon. Neutrophils form a key part of host response to infection. The chemo also adversely affects many mucous membranes and GI mucosa and barriers that are normally resistant to translocation of bugs (bacteria and fungi) are now open. The combination of the bugs in the wrong places and a deficient host response leads to overwhelming sepsis without appropriate treatment. Finally the underlying malignancy itself can interrupt the immune system (think of haematolgical malignancies destroying bone marrow) leading to their own types of infections.
Clinical Presentation
expect to see someone on chemo either mid cycle or recently finished. Given the bewildering number of potential options it’s hard to say when the nadir of neutrophils will be following the most recent chemo. When I first started 7-14 days post chemo was considered a common danger time.
Patients are well educated to be wary of fevers and present if they have one even if they feel well. You might get lucky and they might have actual symptoms pointing you to a source but a lot of the time all you will have is fever.
We often shoot for the low hanging fruit of urine and chest infections but the key take home is to look in unexpected places. 80% of the bugs are thought to be endogenous to the patient.
- mucositis can be an entry point for bugs
- lines
- pressure areas
- biposy sites
- internal metal work (eg biliary stents)
In terms of bugs (adapted from UTD article)
- Gram-negative bacteria (eg, P. aeruginosa) are generally associated with the most serious infections.
- S. epidermidis is the most common gram-positive pathogen, accounting for approximately one-half of all infections due to gram-positive infections. It is much less virulent than other bacterial pathogens
- Although anaerobic bacteria are abundant in the alimentary tract, they are infrequent pathogens isolated from patients with neutropenic fever. However, they can contribute to the pathogenesis of necrotizing mucositis, sinusitis, periodontal cellulitis, perirectal cellulitis, intra-abdominal or pelvic infection, and neutropenic enterocolitis (typhlitis) and can cause anaerobic bacteremia.
- Polymicrobial infections are infrequent, but their frequency appears to be rising
- A bug is only identified in about 20-30% of presentations
Investigation
one of the rare instances where blood cultures are really important. try and get them as clean as possible and consider the other sites like wounds and throats.
usual bloods as you would for any septic patient
The definition of neutrpaenia for our purposes is ANC <0.5 (though anywhere under 2 in context would make me twitchy)
Management
Most of the principles of sepsis apply here.
There will almost certainly be a local antibiotic protocol. NICE recommends piperacillin/tazobactam. It does not recommend routine gent but allows this to be modified locally. Importantly they want us to give the antibiotics if we “suspect” neutropenic sepsis – we don’t have to wait for the ANC result.
I find that almost all of the “suspected” patients will get admitted in Irish practice. Even if the ANC comes back at 1.3 – if they’ve had a fever and chemo they almost all will come in overnight.
Of note NICE does allow for out patient management but there’s lot’s of caveats to it and they have to be very low risk.
There are scores available for risk stratifying them so it’s nice to know they exist but I don’t think they’re currently that relevant to local practice. The MASCC (not that one…) or the CISNE score are the key ones to remember.
The one thing to consider is fungal infections. They’re much less common that bacterial. We should think about them in the highest risk patients (stem cell transplant is a good example) and indeed the MASCC score can be used to risk stratify people to the highest risk.
If you’re seeing someone with recurring neutropenic sepsis then the best tips would be to check what bugs they grew last time and again consider antifungals as recurrent neutropenic sepsis should make you think of it.
Viruses are common in the highest risks patients but they are usually already on prophylaxis.
SVC Syndrome
this is rare (I think i’ve seen it once) but very cool in terms of its presentation and pathophysiology. I covered it once before and reproduced it here.
Two main causes
- malignancy 80%
- thrombosis 20%
For malignancy think right sided lung and mediastinal masses. Anything that can compress the SVC as it returns to the right atrium
Classically this will be non small cell lung cancer followed by non Hodgkin lymphomas
For thrombotic causes then there’s usually a precipitant in the form of an indwelling line
- dialysis, port, Hickman, pacer etc…
- this is the same as for upper limb DVT in general – there’s usually a cause.
A big clot in the major veins returning to the heart would be expected to be a big cause of PE but this seems uncommon. There also seems to be a differentiation between clots on the line (sleeve thrombi) which more commonly cause PE, as opposed to clot on the wall (mural thrombus) which embolises much more rarely.
Presentation
- dyspnoea the commonest (presumably a mixture of reduced venous return and neck vein distension)
- usually there are facial changes – swelling, redness, especially with bending forwards
- Pemberton’s sign is redness in the face on raising the arms, presumably congestion with increased venous return from the arms.
Investigations
- CXR usually abnormal (though up to date quoted 85% only)
- ultrasound can usually only view the vessels immediately proximal to the SVC but you can get indirect information by looking at Doppler waveforms with a valsalva (you were all doing that already for your DVT scans weren’t you…)
- unsurprisingly CT is the best test as the key treatment decision is finding out whether this is a malignant SVC obstruction vs a thrombotic SVC syndrome
- the next major treatment decision point is biopsy if malignant. The decisions regarding radio and chemo that the oncologist will be driven by this.
I mention the last point to emphasise that while this is an important cancer emergency the time scale is quite different from something like neutropenia sepsis.
In health systems where access to CT at 2am is limited then this is a CT that can wait. SVC is rarely an immediately life threatening emergency and intervention (such as stenting, radio, biopsy and chemo) can all wait till daylight and as a result the CT can probably wait till morning too.
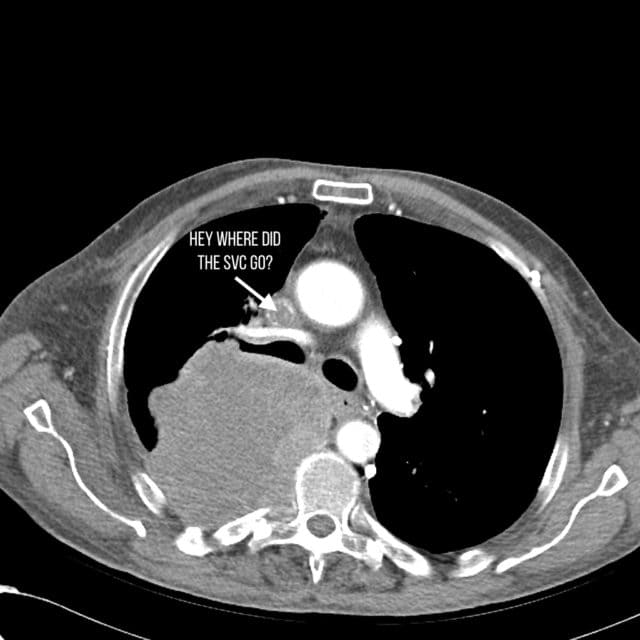
Case courtesy of Dr Henry Knipe, Radiopaedia.org. From the case rID: 28046
Hypercalcaemia
This is common, and also something covered here before.
This is commonly picked up because the person before you randomly ordered some tests and this came back high. Occasionally you might actually suspect it but mostly you’ll just be fishing. The symptoms are typically quite non specific so it’s always worth considering.
Levels about 3 are when it becomes symptomatic and interesting. 2.5-3.0 are still important in terms of a diagnosis but are unlikely to make anyone sick
Once you have a high Ca then consider causes
Malignancies
- myeloma
- breast
- renal
- lung
A lot of this is due to bone mets which makes sense, however most have no bone mets and even in those with bone mets the commonest mechanism of hyper Ca is known as Humoral hypercalcaemia and from the tumour in question producing “parathyroid hormone related peptide”
Other causes
- hyperparathyroidism
- paraneoplastic syndromes
- sarcoid
- addisson’s
- dehydration
- drugs
- lithium
- thiazides
- prolonged cuff time when measuring
There are some ECG changes (which are probably irrelevant but they’d be good as an exam question
- QT shortening (of note in hypocalcaemia the QT gets longer but that’s about the only ECG change
- J waves (osborne waves) can happen
- eventually can have ventricular arrhthmia
- widening of the QRS
Management
from our point of view it’s primarily fluids. bisphosphonates, calcitonin and diruetics probably all have a role but in the timescale we’re interested fluids and a catheter and good fluid balance is all we need to do.
Cord compression
Hopefully you all know that a history of cancer is a red flag in a back pain presentation. This can be quite a remote history especially if it’s breast cancer where recurrences 10-15 years after treatment are fairly common.
Lung, kidney, breast, prostate and myeloma are common causes here.
Back pain is obviously a common presentation but radicular symptoms and lower limb or bladder issues should also prompt you thinking about it. Any time you have a possible cauda equina patient you should consider malinancy. You might get lucky and see a lesion or a collapse on an xray but ultimately you’re gonna need MRI to see the cord. Be sure to MRI the whole core as multiple levels may well be involved.
The thoracic spine has the most number of vertebra and highest blood supply and therefore gets the most mets followed by the lumbar. You can get mets to the meninges and the cord itself but >90% are external compression from a bony met or collapse.
There’s definitely a role for “emergency” radiotherapy here but be realistic – that means radio within a week… Just because it’s cancer don’t skip orthopaedic invovlement as some of them are grossly unstable needing surgical fixation.
As with any time there’s CNS oedema (think brain mets) then steroids probably have a role. Though by the time you’ve got diagnosis with MRI I suspect someone else will be making that decision.
Further Reading
- EM Cases
- REBEL EM
- EMDOCS
- Rosen’s 8th Chapter 123
- NICE Guidance
